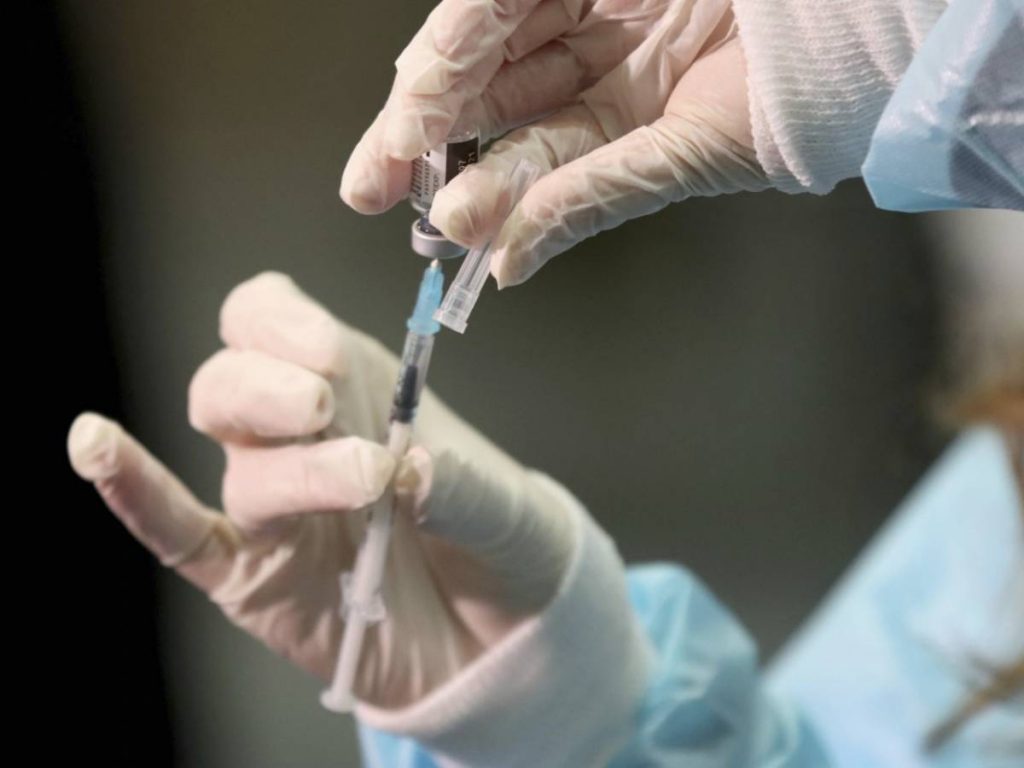
AIFA’s green light for the heterologous vaccine, as expected, has unleashed another controversy over the administration in our country. The vaccine campaign must continue, vaccination Astrogeneca For subjects under the age of 60 it forced them to reconsider the road map completely, but it seems that the square has been found to overcome this obstacle. But not everyone agrees on the management Mix the vaccine People under the age of 60 received AstraZeneca as their first dose. While one section of the scientific community supports this decision and talks about the high efficacy of vaccines, another section is skeptical and reluctant and proposes free selection for vaccines.
Who is against mixing
The true validity of the heterologous vaccine is highly questionable Massimo Andreoni, Scientific Director of the Italian Society of Infectious and Tropical Diseases. The doctor, who was interviewed by Adkronos, expressed some doubts about the path taken by the pharmaceutical company, in his opinion, “This went a little behind the events rather than scientific approaches: expect more data from ongoing studies. I understand that we are in a ‘battle’ against the cove, but some choices will have repercussions. It seems to me that this is a concept pulled by the hair“. A clear comment from Andrioni shows more than a few doubts before the possibility of giving Pfizer or Moderna to those who received the first dose of astrogen.
Andreoni, head of the Dor Vercata Polyclinic in Rome, explains: “Now the situation is more complicated. I fear that this choice may create a ‘hole’ in the vaccine campaign, as there is a risk of confusion and undoubted increase in those who have already taken the first dose of astrogen. Maybe it’s better to get out of there Freedom of choice Allow these individuals to take a second dose as well“
He has a different perspective, but not the opposite, Andrea Chrysanthemum. The director of the Department of Molecular Medicine at the University of Padua, who was interviewed on microphones one day as a goat,From a theoretical, immunological and biochemical point of view there are no elements that suggest the compound is harmful.“However, as he explains later, it does not mean that we have studies that prove otherwise:”It may be more immune, but we will make it clear that we are doing something without being judged by regulatory authorities. There is a lack of studies, we do not have the comfort of data, in some ways this is a test thing“So, the professor underscores that again.”However, AstraZeneca is a safe vaccine, so do not panic if you do. Pfizer and Moderna have fewer side effects“.
He echoes him Maria Rita Kismondo, Director of the Laboratory of Clinical Microbiology, Virology and Biomedical Emergency Diagnosis at Sacco Hospital in Milan. “This does not seem to be based on solid scientific evidence“Kismondo admitted to Adkronos that he was not comfortable with the vaccine in terms of performance:”I will be very relaxed about the side effects“.
Who support the mix
Still different Pierreloki Lopalko, Puglia Region Epidemiologist and Health Councilor: “Unfortunately, in an emergency, health policy decisions must also be made on the basis of weak (non-existent) evidence. The same rules of normal health administration cannot be applied in an epidemic“According to the Commissioner, proper communication is fundamental at every level.”Explain well to the citizen the reason for some choices“About vaccines.
The virologist is also betting on a political decision Fabrizio Bregliasco, Sees this as an option in Atnchronos “To reassure the spirits, with the utmost hope of moving the vaccination campaign forward“Compared to his peers, Bregliasco is more confident in this conclusion:” CThere are scientific elements, however. Other countries have done it, there is data“.
Matteo Pacetti The positive one is: “The combination of vaccines is recognized in other countries such as Canada and Germany: it is not always a question of saying it should be done this way, but in Italy if you change the target of one vaccine you can do a second dose with a different vaccine. Studies, it seems to me, can do that, albeit preliminary. Change is appropriate if the scientific evidence in medicine changes“.
Further Pierre Mannusio Mannusi, Hematologist and AIFA consultant on the issue of astrogenica, supports the heterologous vaccine: “The science and technology team performed well. It is true that not much data is available on the background of the Govt vaccine. But we have become accustomed to changing the type of vaccine frequently in diseases such as measles and flu. Preliminary reports suggest that the compound is effective and has no safety concerns. I understand who says: we need more data. But they will come soon“.
This decision was also claimed Nicola Magrini, IFA Director General: “We need to be more than just being quiet. At this stage of the epidemic, young people were chosen to avoid being vaccinated at the most distant risk of a serious event: everyone has the privilege of being guaranteed the safest vaccines possible. The message to be given is hope and maximum peace of mind“.
Therefore, he also had his opinion on the matter Pierpolo Celery: “Converting the second dose of AstraZeneca to the M-Arna vaccine is by no means experimental. It is true that there are not many studies, but this is something that is done in many states, perhaps by a third dose in a few months.So the Deputy Secretary, Andrea Krishanthi, responded indirectly.

“Gamer. Professional beer expert. Food specialist. Hardcore zombie geek. Web ninja. Troublemaker.”






More Stories
A Chinese outpost a stone's throw from the US: the latest secret revelation
Romano Prodi: Mine's not an attack on Schlein, but it's better not to have a name on the symbol
Snow, low elevation, will fall in affected areas from Tuesday onwards