The researchers’ analysis found how the virus evolved to increase its transmissibility and evasion
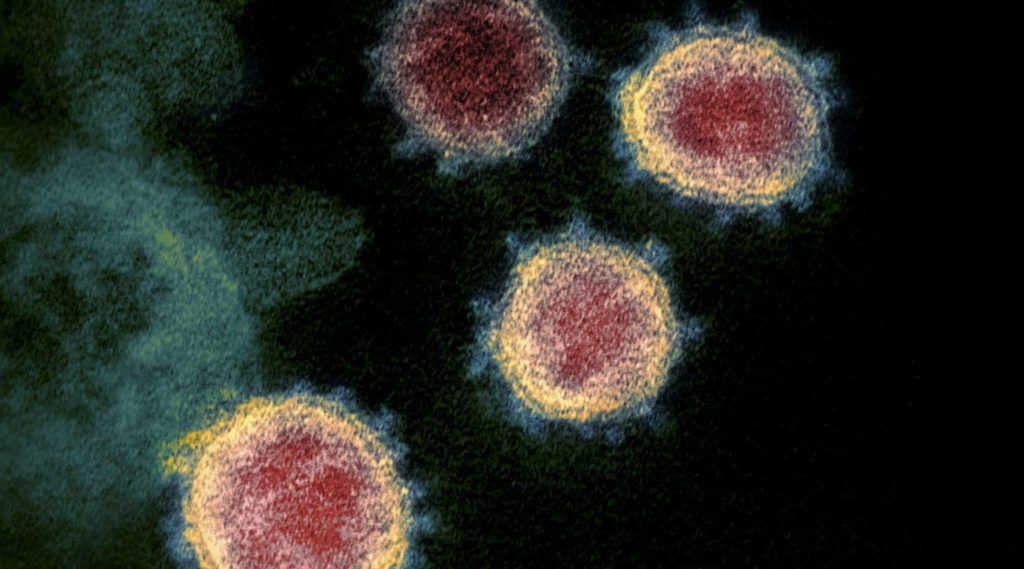
Features of Omicron Spike Protein 37 unpublished mutation, a number three to five times higher than the number observed in the previous variables. Structural analysis, performed thanks to cryo-electron microscopy, indicates that different mutations (R493, S496 and R498) give rise to new ionic bonds and hydrogen bonds between Spike and the Ace-2 receptor. these relationships They increase the affinity of the virus for human cellsWhile other mutations (eg K417N) reduce the strength of these bonds.
“Overall, these results indicate that Omicron has higher binding affinity Compared to the original virus, levels are more similar to what we see with the delta variant,” explains study coordinator, Sriram Subramaniam. It should be noted how the variable OmicronThey have evolved to retain their ability to bind to human cells Despite so many mutations.”
Additional experiments show that Omicron has a greater ability to evade antibodies than previous variants: it is able to evade all six monoclonal organisms tested (five of which are completely ineffective), while also showing an increased ability to evade people-produced antibodies. who have been vaccinated or cured. from Covid-19. “Especially, Omicron escapes vaccine-induced immunity Compared to that caused by natural infection in unvaccinated patients. This – concludes biochemist Subramaniam – indicates that Vaccination remains one of our best defenses“.

“Internet trailblazer. Travelaholic. Passionate social media evangelist. Tv advocate.”






More Stories
The LEGO 10341 NASA Artemis Space Launch System isn't the first of its kind
12 out of 20 regions do not guarantee basic levels
Watch the real video of the probe's descent to Titan (more than 1.3 billion kilometers from Earth)